Lado Healing Institute
Lado Healing Institute
Welcome to Lado Healing Institute
Welcome to my practice’s website! I’m Dr. Leonard Lado, MD, the Founder and Medical Director of Lado Healing Institute. As a board-certified Psychiatrist, I’ve proudly served Southwest Florida since 2002. Recognizing the unique needs of our community, we’ve maintained our independence to offer more choices and ensure personalized attention, unlike larger treatment centers.
At Lado Healing Institute, we stay at the forefront of advancements, introducing innovative approaches like Genetic Testing, Transcranial Magnetic Testing (TMS), and much more to help with all types of mental health conditions. Addressing the current opioid and fentanyl crisis, we provide suboxone treatment, allowing detox without hospitalization.
Our commitment extends to accessibility, facilitated by cutting-edge software for seamless communication. Beyond patient care, I’ve been an educational leader, providing insights to physicians, nurses, and all types of medical staff. Explore our YouTube channel for access to interviews, educational pieces, and to stay up to date in the world of psychiatry. Joins us at Lado Healing Institute, where our compassionate staff is eager to help you regain and live your life to the fullest.
“Breaking the Stigma on Mental Health.”

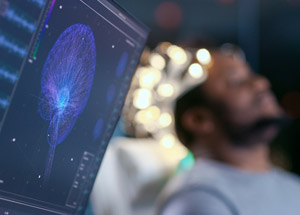
Personalized Genetic Testing
The biggest challenge in the field of psychiatry is simply two-fold. Making the correct psychiatric diagnosis and matching the medications with the person’s metabolism. My practice has the ability to do both Computer Assisted Psychiatric Diagnosis and Pharmacogenetic Testing for Psychotropic Medication response, which takes out the guessing game of matching the right medication for the highly probable diagnosis and patient metabolism. Pharmacogenetic testing is now possible in our facility, and we obtain results within 10-14 days. An excellent web site on how this technology works in our practice is best described by a personalized medicine company AssureRXHealth. Right now, in doctors’ offices all over the world, patients are given medications that either doesn’t work or have harmful side effects. Often, a patient must return to their doctor over and over again until the doctor can find a drug that is right for them. Pharmacogenomics offers a very appealing alternative. Leonard Lado MD now provides a simple and rapid test of your DNA. This gives me the ability to determine if the patients have the possibility of suffering a severe negative reaction to the medication due to side effects. Pharmacogenomic testing is now available in Lee, Collier and Charlotte counties, and right here in our Bonita Springs office serving the entire Fort Myers area.
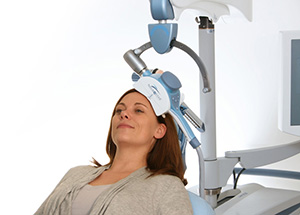
NeuroStar TMS Therapy is the Right Choice
Transcranial magnetic stimulation (TMS) uses a targeted pulsed magnetic field, similar to what is used in an MRI (magnetic resonance imaging) machine. While the patient is awake and alert, NeuroStar TMS Therapy stimulates areas of the brain that are underactive in depression.
Telemedicine & Telepsychiatry
The Lado Healing Institute is proud to provide telemedicine and telepsychiatry technologies to provide psychiatric care over the Internet. Dr. Lado can provide professional services over the Internet. Telepsychiatry and some areas of telemedicine are here to stay. It has many advantages in that a physician can bring his clinical skills to a distant place where help is needed, such as rural areas, correctional and jail systems, and inpatient psychiatric facilities. However, telemedicine is very much limited to areas where data from the patient’s condition can be ascertained through history and in the case of psychiatry the mental status exam. Other new technologies, such as computerized testing to validate and support the psychiatrist diagnosis. The most critical thing I was taught in my training first from surgery, if you don’t know what you’re looking at, how can you treat it. The same applies to psychiatry. If you don’t have an accurate diagnosis, how can you provide effective treatment?